  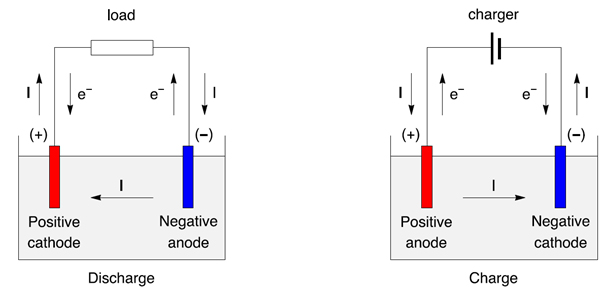
However, spontaneous chemical reactions occur in electrochemical cells, whereas nonspontaneous chemical reactions occur in electrolytic cells. Electrochemical and electrolytic cells both rely on the movement of electrons through the system. As a result, the electrolytic cell has all of the components found in a standard electrochemical cell. In electrochemical cells, electrons go from the anode to the cathodeĬhemical energy is turned into electrical energy in an electrochemical cellĮlectrons go from the battery to the cathode, then via the electrolytic solution to the anode in electrolytic cellsĮlectrical energy is turned into chemical energy in an electrolytic cellĪ type of electrochemical cell is an electrolytic cell. The anode of an electrolytic cell is positive, whereas the cathode is negative The anode of an electrochemical cell is negative, whereas the cathode is positive Nonspontaneous chemical reactions occur in electrolytic cells As a result, the cathode has a negative charge whereas the anode has a positive charge.ĭifference Between Electrochemical Cell and Electrolytic Cell Electrochemical CellĪn electrochemical cell is a system that uses spontaneous chemical reactions to generate electrical energyĪn electrolytic cell is a form of electrochemical cell in which electrical energy is utilised to initiate a chemical reactionĮxternal electrical energy sources are not required for electrochemical cellsĮxternal electrical energy sources are required for electrolytic cellsĬhemical reactions occur spontaneously in electrochemical cells 
These anions then lose electrons and get oxidised. The cathode’s electrons repel the anions in the solution. As a result, at the cathode, these cations are decreased. The cations in the solution then congregate around the cathode and absorb electrons that are passing through it. The electrons are subsequently injected into the electrolytic solution. The anode will undergo the oxidation reaction, whereas the cathode will undergo the reduction reaction.Īn electron flow via the cathode is provided by an external electrical energy supply (from the battery attached to the two electrodes). One electrode serves as an anode, while the other serves as a cathode. An external circuit is linked to two electrodes. Solid metal electrodes are also used in electrolytic cells. The most common type of electrolytic cell used for compound electrolysis is the electrolytic cell. 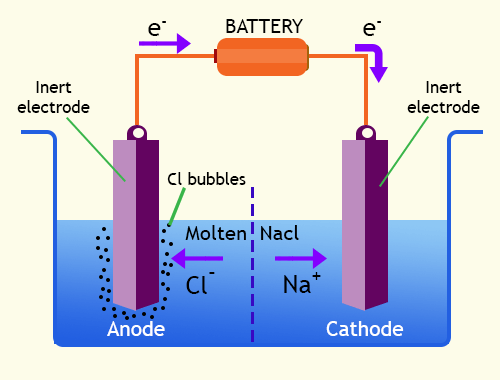
Then you can start a nonspontaneous response. To put it another way, electrical energy should come from somewhere else. An electrolytic cell is a form of an electrochemical cell that uses electrical energy to initiate a chemical reaction. In two half cells, half-reactions take place.Īn electrolytic cell is a device that uses electricity to produce electricity. Two half cells make up an electrochemical cell. Voltaic (galvanic) cells and electrolytic cells are two forms of electrochemical cells. As a result, the two half-reactions happen at the same time. When an oxidation reaction occurs, electrons are always released into the system, but when a reduction reaction occurs, electrons are taken away from the system. A redox reaction is made up of two half-reactions: oxidation and reduction. Electrons are transferred between chemical species in redox reactions. Redox reactions are the chemical processes that take place throughout this process. An electrochemical cell is a device that can generate electrical energy from chemical processes that occur spontaneously. The major distinction between an electrochemical cell and an electrolytic cell is that an electrochemical cell does not require external current to function, but electrolytic cells do.Ĭheck out: CBSE 12th Result 2022 What is an Electrochemical Cell?Īn electrochemical cell is a device that converts electricity into chemical energy. Voltaic cells and electrolytic cells are the two forms of electrochemical cells. Cells, or more accurately, electrochemical cells, are the systems in which these transitions take to happen. Electrical energy will be converted into chemical energy or the other way around in both cases. Chemical reactions can either be employed to create an electrical current or to assist the occurrence of a nonspontaneous chemical reaction. The study of electron transport in systems undergoing chemical reactions is known as electrochemistry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
